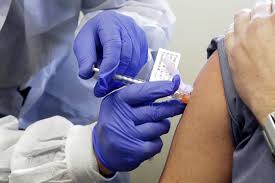
After the vaccine of two companies in the war against the Coronavirus has been approved for emergency use, it may be difficult for the vaccine companies standing in front lines to be tested. There may be a problem with getting volunteers for their vaccines. It is known that on January 3 of this year, DCGI approved the Oxford Vaccine and indigenous Covaxin made at Serum Institute. Companies say recruitment has been adversely affected due to the news of the early availability of the vaccine.
Dr. V Krishna Mohan of Bharat Biotech said in a webinar, "There are several press releases without getting approval for emergency use, saying that their vaccine will be available soon." This is going to cause a lot of difficulty in recruitment, because most people are thinking about whether they should take part in clinical trials or not. After all, the vaccine is going to come soon. " Trials are taking place at the same time when the work of approving vaccines for emergency use is also going on in the country.
'Big challenge for India biotech'
He said that the challenge for Bharat Biotech was huge, as they were focusing on the older age group. He said that the oldest person to enroll for the covariate trial was 97. Mohan said, "Hospitals have also asked us whether it is morally right to continue clinical trials even when you have received emergency use. These questions should be answered. "Let us know that at least six human trials of corona vaccines are going on in the country. This is Zydus Cadila, which has recently started a third phase trial. Russia has Sputnik 5, whose second and third phase trials are being conducted by Dr. Reddy.
There is also an ongoing trial of these vaccines
Serum Institute of India also has the second vaccine Novavax, which is undergoing Phase II trials. Biological E and Genova are testing the first phase of their vaccine. Apart from this, work is also underway on another biotech vaccine of Bharat Biotech. Cadila Healthcare Chairperson Dr. Pankaj Patel also believes that recruitment time is going to be very challenging. He says the challenge will be to meet recruitment speed and volunteers. At the same time, there is also a concern that after the introduction of the vaccine, the weak people will get vaccinated soon.
Pakistan cricket team captain Babar Azam has refused to endorse and wear apparel bearing the logo
Maharashtra: 'Those who are glorifying Aurangzeb are traitors', statement of Deputy CM Eknath Shinde
Maharashtra Deputy Chief Minister Eknath Shinde has said that those who are still praising Aurang
The 68th match of IPL 2024 between Royal Challengers Bangalore and Chennai Super Kings will be pl
Afghan women raised flags in support of Iran, Taliban fighters fired upon them
The process of protests in Iran over the death of a woman during police custody in Iran is not ta
Elon Musk followed PM Modi on Twitter, users are giving this reaction
Twitter chief and billionaire Elon Musk has started following Prime Minister Narendra Modi on Twi
Amitabh-Ravi: Ravi Kishan praised Amitabh Bachchan for 'Kalki 2898 AD', Big B greeted him with a hug
Amitabh Bachchan is currently enjoying the success of his recently released film 'Kalki 2898 AD'.
The Delhi High Court will pronounce its verdict on Kejriwal's plea seeking a stay on the trial co
If you too are planning to travel by train during this summer vacation and are worried about the
Ukraine has now sought India's support to end the war with Russia. Ukraine's President's Office c
Sri Lankan great spinner Muttiah Muralitharan says current Indian coach Rahul Dravid is included